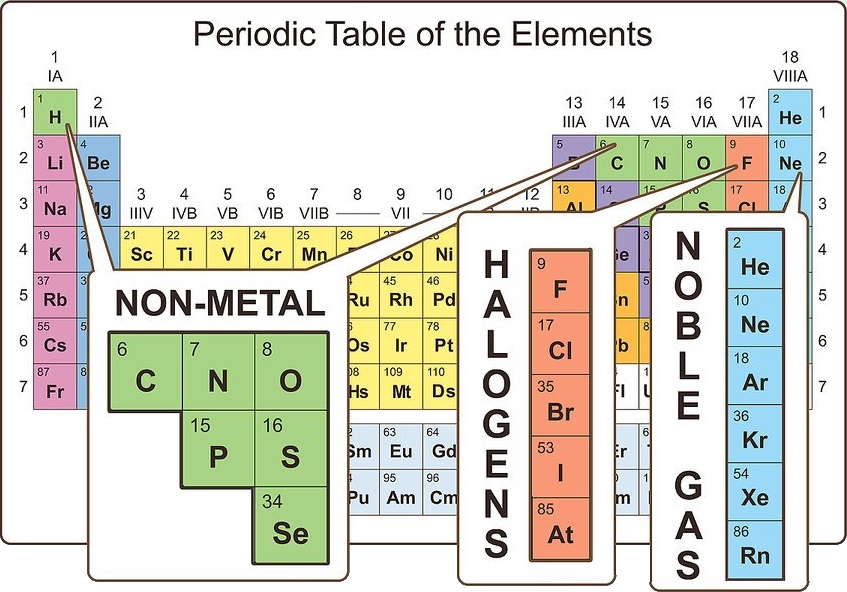
You should note the pattern here compares with the layout of the periodic table. However, their chemical and physical properties are dramatically different. The standard atomic mass of carbon is 12.0096 and its symbol is ‘C’. Lead and carbon are part of the same group on the periodic table, so they have the same number of valence electrons. All neutral elements will have the same number of protons and electrons, which is equal to the atomic number. First we determine the number of electrons in the atom then we add electrons one at a time to the lowest-energy orbital available without violating the Pauli principle.We use the orbital energy diagram of Figure 2.1. Carbon is the 6th element in the periodic table. 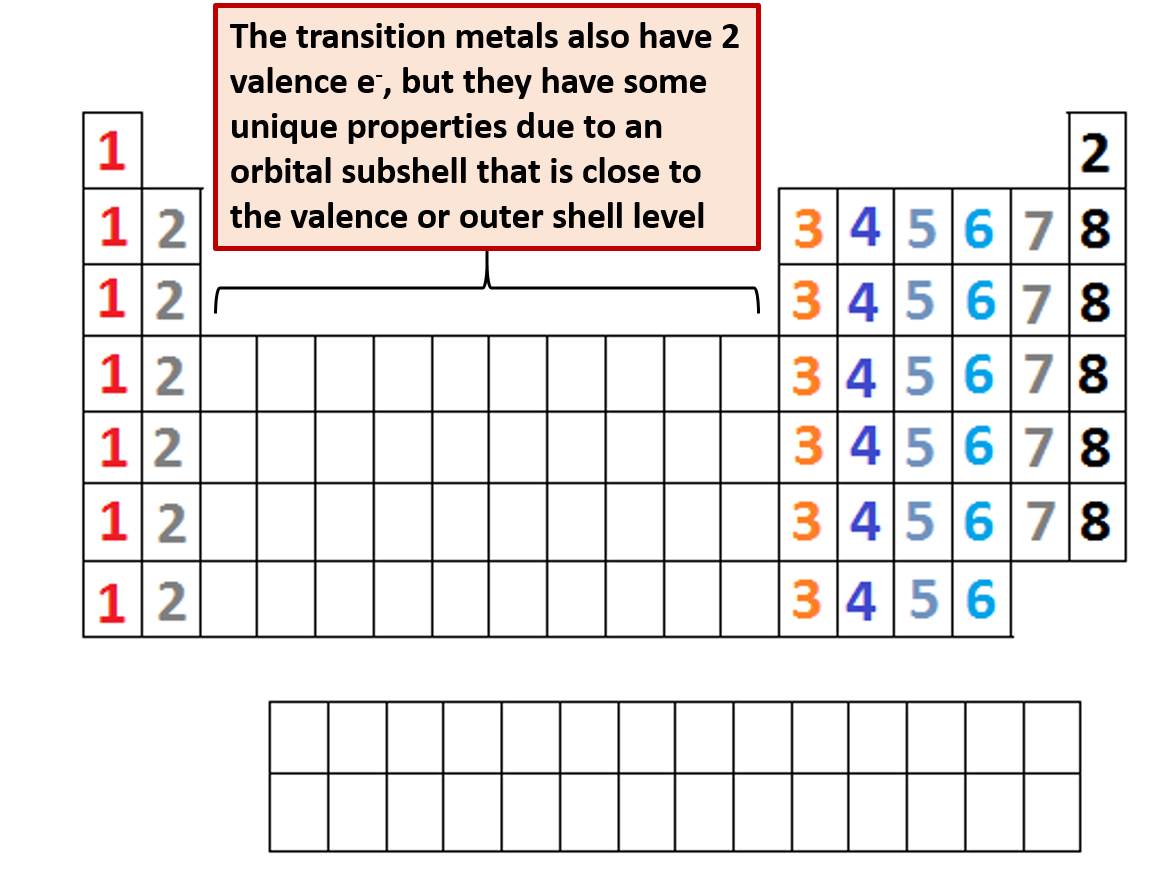
The usual valency values of some elements are shown in Table 2. Carbon is the sixth element in the periodic table and the 1st element in group-14. In general, the number of valence electrons is the same within a column and increases from left to right within a row. We construct the periodic table by following the aufbau principle (from German, meaning building up). This tendency for atoms to behave as though they have a specific number of ‘hooks’ is known as valency, and is sometimes referred to as the combining power of an atom (or a group of atoms). Similarly the chlorine atom also has one ‘hook’, so the prediction for the formula of the compound from carbon and chlorine is CCl 4. However, the most common method uses atom’s ground state electron configuration. (i.e. The easiest way to find the number of valence electrons is to go by the element group in the valence periodic table. The simplest compound of carbon and hydrogen is the gas methane, CH 4: a formula that would be obtained if it were assumed that carbon had four ‘hooks’ and hydrogen had one. Carbon group number marked on periodic table The valence electrons of each main-group element can be determined by the column in which it is located. Here the number of valence electrons corresponds directly to the number of hooks, the link is not always so simple, as you will see. Here the number of valence electrons corresponds directly to the number of ‘hooks’. So, in the valence shell (in this case corresponding to the principal quantum number 2), there are four electrons. The electronic configuration of the carbon atom is 1s 2 2s 2 2p 2. How many valence electrons does carbon have?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
